|
It reacts with sulfur, and forming sodium sulfide: When Sodium reacts with hydrogen, it forms sodium hydride: The metal dissolves readily in dilute sulphuric acid (H 2SO 4), and forming the solutions containing the aquated Na (I) ion together with hydrogen gas (H 2).Ģ Na (s) + H 2SO 4 (aq) → 2 Na + (aq) + SO 4 2- (aq) + H 2 (g) It reacts vigorously with Halogens, and forming the corresponding sodium (I) halides:Ģ Na (s) + F 2 (g) → 2 NaF (s) (Sodium fluoride)Ģ Na (s) + Cl 2 (g) → 2 NaCl (s) (Sodium chloride)Ģ Na (s) + Br 2 (g) → 2 NaBr (s) (Sodium bromide)Ģ Na (s) + I 2 (g) → 2 NaI (s) (Sodium iodide) It is very exothermic reaction, where during the reaction, the metal become so hot, and due to high temprature, it catches fire and burn with a orange color:Ģ Na (s) + 2 H 2O → 2 NaOH (aq) + H 2 (g)Ģ Na (s) + 2 H 2O (l) → 2 Na + (aq) + 2 OH ‑ (aq) + H 2 (g)  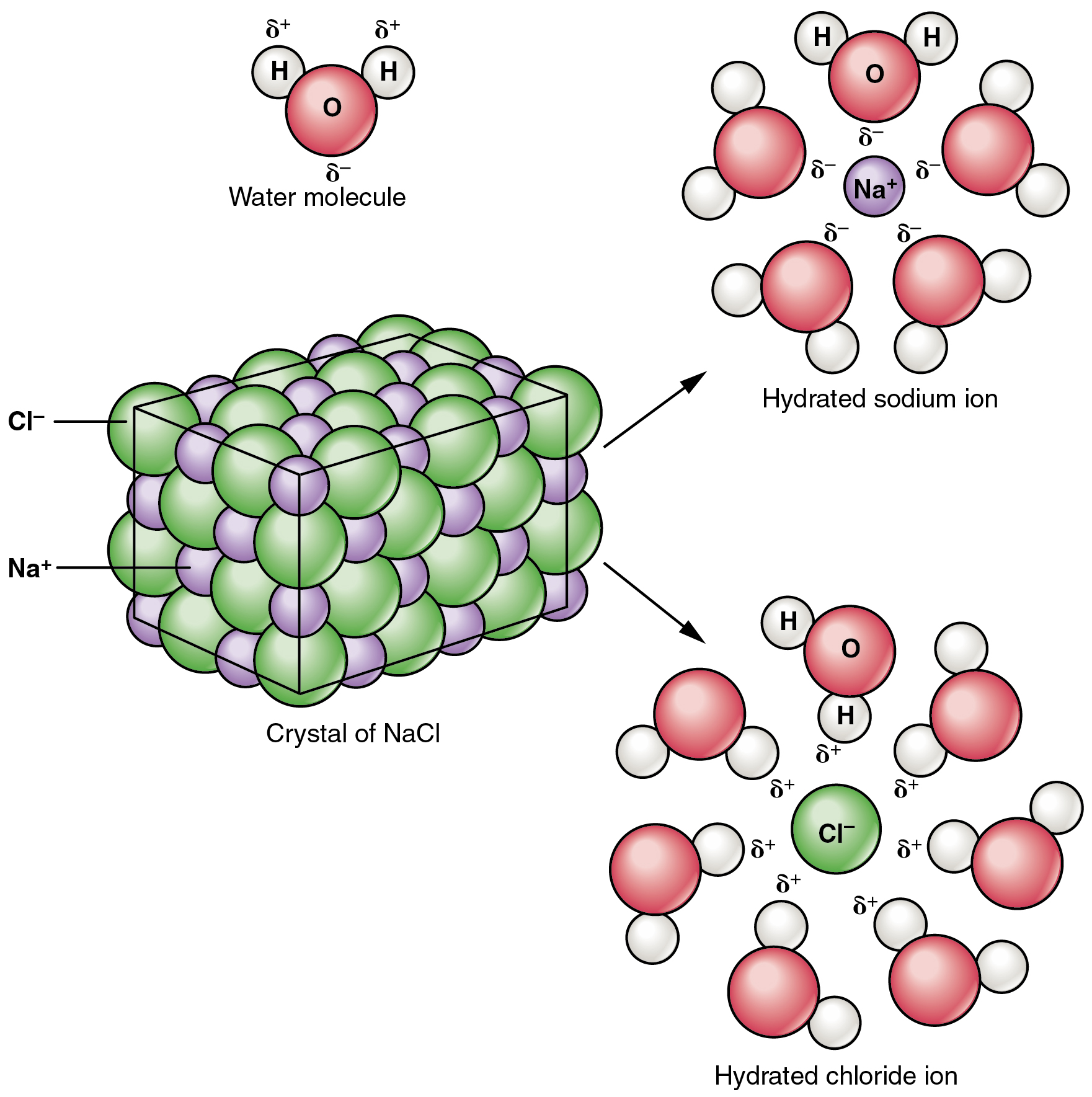
The metal reacts violently with water, where it forms a colorless solution of sodium hydroxide (NaOH) & Haydrogen (H 2). Sodium reacts with air, and forming sodium oxide (Na 2O), But if sodium burned, than it forms sodium peroxide (Na 2O 2) with some sodium oxide: Lattice constant: 428.20, 428.20, 428.20 pmīody Centered Cubic (BCC) Reactivity of Sodium Ionization energies: 1st: 495.70 kJ.mol 2nd: 4561.5 kJ/mol 3rd: 6910 kJ/molĬrystal structure: Body centered cubic (B.C.C) Molar magnetic susceptibility: 0.2×10 -9 m 3/mol Physical Properties of Sodiumĭensity: 0.967 g/cm 3 (In solid) 0.928 g/cm 3 (In Liquid at M.P) Mass magnetic susceptibility: 6.4×10 -9 m 3/kg 
Volume magnetic susceptibility: 0.0000062 Magnetic susceptibility (x mol): +16×10 -6 cm 3/mol Thermal conductivity: 142 W/(m∙K) Electrical properties of SodiumĪ Electrical type: Conductor Magnetic Properties of Sodium Sodium reacts with alcohols is similar, as reacts with water, But with alcohol, it reacts slowly. That’s why the metal stores in Kerosene (a complex mixture of Naphthenes (40.8%), Paraffins (55.3%), and Aromatic hydrocarbons (3.9%). Sodium doesn’t react with paraffinic hydrocarbons (alkane), But it forms addition compounds with naphthalene (C 10H 8), aryl alkenes, and other aromatic polycyclic compounds. It also reacts with various metallic halides to form the metal & sodium chloride (NaCl). Sodium rarely reacts with carbon, but it does react with halogens (F, Cl, Br, I). Sodium & hydrogen reacts above 200✬ to form sodium hydride (NaH). Sodium doesn’t react with nitrogen, even not react at very high temperatures, but it can react with ammonia (NH 3) to form sodium amide (NaNH 2). 
It may or may not ignite spontaneously on water, It all depends on the amount of oxide and metal exposed to the water,Ī tiny amount of metal floats on water, but not only high, even normal quantity of metal ignite spontaneously on water. Sodium reacts readily with water, snow & ice, to produce sodium hydroxide (NaOH) and hydrogen. Sodium metal reacts with air, and looses its silvery appearance and acquires an opaque (not transparent ) grey color due to the formation of a sodium oxide (Na 2O) coating. Sodium is a soft, reactive, bright-silvery metal with the low melting point, and it is not found free in nature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
